
The CMTA looks forward to a time when doctors are able to use genetic therapies to treat the root cause of CMT rather than prescribing medications or recommending surgery. We are already envisioning the possibilities that gene therapy holds for our community of 2.8 million people worldwide living with CMT. In fact, we’re leading the pursuit to explore gene therapy in CMT by expanding our Strategy to Accelerate Research (STAR) program and our STAR Advisory Board.
In CMT, the genetic causes for many types of CMT are already known, which allows scientists to more efficiently zero in on genetic solutions to treat—and possibly cure—the disease. As we continue gene therapy research inside the lab, our ultimate goal is to transform it into effective treatments for people living with CMT.
Given the increased feasibility and applicability of gene therapy to CMT, the CMTA hosted a Gene Therapy Workshop in 2018. In response to invitations from CMTA board member Dr. Steven Scherer, more than 20 of the top gene therapy experts gathered for the inaugural CMT-centered workshop on gene therapy. This meeting included experts who have worked in related genetic and neuromuscular disease areas, as well as clinicians and scientists spearheading efforts toward gene therapy for CMT2D and CMT4J.
Building on this meeting, the CMTA added several experts to the Scientific Advisory Board to formulate gene therapy strategies for CMT2 and CMT1 subtypes. These include Kleopas Kleopa, MD, at the Cyprus Institute of Neurology & Genetics, Scott Harper, PhD, at the Ohio State University School of Medicine, and Steven Gray, PhD, at the University of Texas Southwestern Medical Center, all of whom have already initiated projects devoted to gene therapies for CMT. Dr. Kleopa is a neuromuscular neurologist and has shown proof of concept that gene therapy works in two mouse models of CMT: CMT1X and CMT4C. He is also extending his efforts to develop novel therapies for CMT1A. Dr. Harper has worked extensively in muscular dystrophy and is collaborating with Robert Burgess, PhD, at the Jackson Laboratory to develop a gene therapy treatment for CMT2D. Dr. Gray’s core expertise is in Adeno-Associated Virus (AAV) gene therapy vector engineering, followed by optimizing approaches to deliver a gene to the nervous system. He is leading efforts to develop novel gene therapy for two types of CMT: GAN and CMT4J.
WHAT IS GENE THERAPY?
Our genes dictate many of our personal characteristics; however, mutations in genes cause genetic diseases, such as CMT. Scientists have been working for decades to modify or replace faulty genes with healthy ones to treat, cure or prevent disease. Fortunately, we are seeing significant progress on these efforts to provide gene therapy options for CMT. In fact, recent studies have provided an effective gene therapy for spinal muscular atrophy (SMA), a devastating disorder that affects the same motor neurons that are affected by CMT.
GENE EDITING AND GENE REPLACEMENT
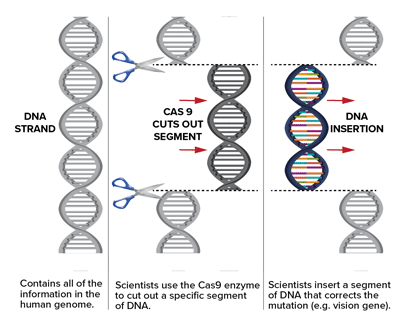
Sometimes the whole gene is duplicated, as in CMT1A, where a chromosome segment around the PMP22 gene is present in three copies instead of two. Alternatively, a part of a gene is defective or missing from birth, causing many of the other known forms of CMT. Any of these variations can disrupt the structure of the protein that is encoded by the affected gene, causing cellular problems that ultimately lead to disease.
In gene therapy, scientists can do one of several things depending on the problem with the gene. The simplest form of gene therapy is to simply provide a correct copy of the gene, which is the basis of the gene therapy for SMA. In variations of this approach, genes that are causing problems can be suppressed. One example of this was the recent demonstration that antisense oligonucleotides can be used to improve the neuropathy in rodent models of CMT1A. In addition, the exciting new field of genome editing using CRISPR technology has now made it possible to correct disease-causing mutations, and collaborative projects have already been initiated with leaders in this field.
GENE THERAPY DELIVERY
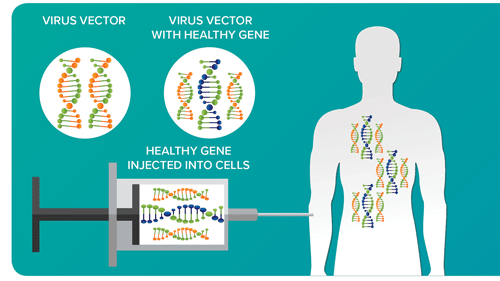
In order to insert new genes directly into cells, scientists use a vehicle called a vector that is genetically engineered to deliver the correct version of the gene. For example, viruses have a natural ability to deliver genetic material into cells, and therefore, can be used as vectors. While some viruses cause disease, virus vectors are highly modified to remove their ability to cause disease so that they can be safely used to carry therapeutic genes into human cells.
Gene therapy can be used to modify cells inside or outside the body. When it’s done inside the body, a doctor will inject the vector carrying the gene directly into the part of the body that has defective cells.
ACCELERATING GENE THERAPY RESEARCH
Before a company can market a gene therapy product for use in humans, the gene therapy product has to be tested for safety and effectiveness so that the Food and Drug Administration (FDA) can evaluate whether the risks of the therapy are acceptable in light of its potential benefits. Gene therapies have begun to receive FDA approval, and many gene therapies are in clinical trials.
At the CMTA, we believe gene therapy holds the promise to provide effective therapies for people living with CMT. As we continue to make great strides in this area, the CMTA is committed to helping speed the development of gene therapy approaches by investing in the most promising and groundbreaking gene therapy treatments that have the potential to benefit our community.
ANSWERS TO GENE THERAPY QUESTIONS
We are members of the National Organization for Rare Disorders (NORD), and they have put together a six-minute video to help answer questions frequently asked about gene therapy. We think this video will help you better understand the basics of gene therapy.

Read about the Patients as Partners in Research Initiative ⟶





